Hot STM Labs
![]()
Chemical Vapor Deposition of Dimethylaluminum Hydride on the Si(001) Surface
Experimental
Results
Room Temperature Dose - Adsorption
Configurations
Annealing R.T. Dose - Conversions and
Decomposition
Dosing above Room Temperature
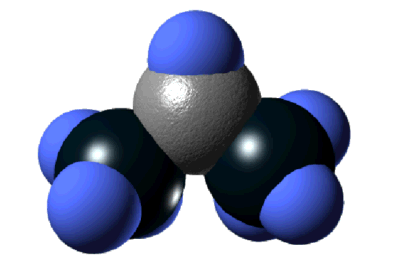
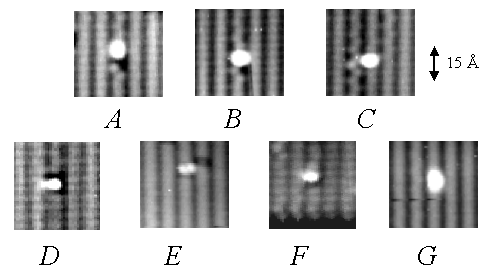
Figure 1. STM images of the seven DMAH adsorption features observed upon dosing a room temperature Si(001) surface. The distinguishing characteristics of these features include differences in the bright spots’ position across and along the dimer rows, induced substrate dimer buckling, the presence of dark imaging substrate dimers, and the presence of substrate dangling bonds. Filled state images, –2.0 V bias.

FIG. 2. A set of STM images of the Si(001) surface (a) before and (b) after two adsorptions of DMAH in configuration G, and (c) after one of these has desorbed or hopped from its adsorption site and the other has converted to the more stable configuration E. The reverse of this process infrequently observed. Filled state images, -2.0 V bias.
The observation of state conversions supports the identification of the seven features with different configurations of a lesser number of species. Immediately after dosing, configuration G has been observed. The DMAH species in this state either diffuses or converts into configuration E. Both cases are shown in Fig.2. The reverse of this process has also been observed. Employing high sample biases, near -2.5 V, while scanning has induced other conversions between states. These include reversible conversions between configurations A and D with the common dark dimer (not present before adsorption) remaining stationary, and a conversion from configuration E to B. From this, we can conclude that features A and D are produced by the same species of DMAH, and E, B, and G are produced by the same DMAH species.

FIG. 3. Before and after images of a tip dissociated B configuration. The feature in (a) and (b) is identified as a B-type DMAH adsorption feature. The tip is positioned over the feature and a +7 V sample pulse is applied. This produces a pair of Al monomers (c), which convert to an Al dimer (d) and (e) within 1 minute. The sample biases employed during imaging are shown on the individual images.
To determine the DMAH species (monomer, dimer, or trimer) responsible for the different features, we employ tip induced molecular dissociation.16 This approach is illustrated in Fig. 3. The feature B is identified in Fig. 3 (a) and (b) under different bias conditions. The tip is positioned over the feature and a 7 V pulse is applied to the sample. Immediately after the pulse, two Al monomers are seen, Fig. 3 (c), which subsequently form an Al dimer, Fig. 3 (d) and (e). Both Al monomers and dimers on the Si(001) surface have been investigated experimentally and theoretically.3,10 Our identification of the two 3 Å protrusions in Fig. 3 (c) as monomers and the feature in Fig. 3 (d) and (e) as a dimer agrees with these works.
Pulsing the sample bias with the tip positioned over DMAH adsorption configurations A and D has produced pairs of Al monomers or single Al dimers in 7 out of 15 cases. Of the remaining tip induced dissociations, in 3 cases single Al monomers were observed, and in the other 5 cases no aluminum containing species were identified. In all 4 cases of induced B-type dissociation, single Al dimers or pairs of Al monomers were produced. It is important to note that the Al monomer is extremely mobile on the Si(001) surface; therefore, only Al monomers trapped by defects or other dissociation fragments were observed at the dissociation site. The features A, B, and D appear to be produced by species containing 2 Al atoms.
This preliminary tip induced dissociation investigation indicates that adsorption features A, B, and D are DMAH dimers. Because conversions have been observed between these features and features E and G, this identification may be extended to these features as well. We believe that all seven of the adsorption features are DMAH dimers. That DMAH dimers dominate the surface, in contrast to the one to one ratio of dimers and trimers in gas phase, may reflect a large difference in dimer and trimer room temperature sticking coefficients or the dissociation of trimers prior to stable adsorption. Theoretical work on both this difference in sticking coefficients and the DMAH dimer bonding configurations are required for a better understanding of these observations.
Annealing R. T. Dose -Conversions and Decomposition
Annealing to 525 K induces state conversions and decomposition. After a 1 min anneal, the density of DMAH configurations is reduced by 30% and only configurations A, D, and F are observed to remain. The ratio of these configuration is D:A:F = 16:3:1. Considering features A and D to be produced by the same species, and observing that the density of A's plus the density of D's does not change due to the anneal, we propose that the change in their relative populations is due entirely to the conversion of 60% of the species in configuration A before the anneal to configuration D after it. Figure 4 shows these configurations and three new features that we identify as the primary DMAH decomposition fragments: H, CH3, and Al.17, 18

FIG. 4. STM image of the Si(001) surface after annealing to 525 K. Only configurations A, E, and F are still present. The three new features are produced by H, Al, and CH3 (usually paired). At high biases F is induced to toggle. Filled state image, -2 V bias.
Figure 4 shows these configurations and three new features which we identify as the primary DMAH decomposition fragments: H, CH3, and Al. Hydrogen on the Si(001) surface has been identified in a number of other works.1,2 Based on the bias dependence of the images, we believe the aluminum feature is an aluminum monomer trapped at a C-type defect. This is consistent with modeling and observations of aluminum monomers on the Si(001) surface.3,4 The C-type defects reappear after annealing to 575 K, and the aluminum forms dilute dimers.
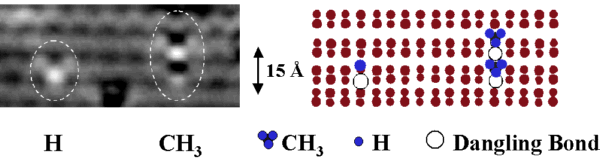
FIG. 5. STM image of H and CH3 adsorption features and corresponding atomic models. The three hydrogens in the CH3 model are arbitrarily placed. Filled state image, -1.5 V bias.
CH3 adsorption on the Si(001) surface has been the subject of both experimental and theoretical works.5,6,7,8 Figure 5 presents an image of the CH3 and a H adsorption and tentative atomic models. The CH3 features are usually paired across two adjacent dimer rows. We believe that the CH3 pairs are located at the site of molecular decomposition.
Dosing on a surface at 350 K, we observe decomposition of the DMAH molecules. After a dose equal to that at room temperature (section A), we observe the primary decomposition fragments (H, CH3, and Al) and a reduced density of the DMAH configurations previously identified. Only configurations A and D are represented. New features, which out number configurations A and D by 9 to 1 are also observed. These features are smaller than those of the seven configurations, and upon annealing the surface to 525 K, these new features are replaced by an equal density of Al atoms, ordered into dilute dimer rows,9,10,11 and an increased density of CH3 and H. Meanwhile, the density of DMAH in configurations A and D remains constant through the anneal. We identify the new species as Al containing DMAH fragments.
We conclude that there are two adsorption paths. One is non-dissociative adsorption producing the configurations A and D, which are stable against decomposition. The other is higher energy dissociative adsorption producing the Al containing fragments, and leading to Al dimer formation upon further annealing.
 |
FIG. 6. (a) STM image of the Si(001) surface after exposure to DMAH at 575 K. (b) Surface after subsequent exposure to three times as much DMAH. Bright spots are aluminum dimers; dim spots are H or CH3. Empty state image, +2 V bias.
Cleaner aluminum growth is achieved by dosing at higher temperatures. At 400 K, none of the Al containing fragments identified at 350 K are observed, and there is a negligible density of the room temperature features A and D, with only one or two per 400 × 400 Å2 image. The surface is otherwise populated with Al, CH3, and H. Our cleanest growth was achieved at 575 K, as shown in Fig. 6. Only Al, CH3, and H are observed. The bright features in this image are Al dimers, primarily arranged in dilute dimer rows. Comparison of Al grown by CVD and by Al evaporation reveals that the rows grown by CVD tend to be much shorter and to terminate at CH3, H, or defects.10,12 The ratio of Al to CH3 plus H is 4 to 1. After a higher dose, Al rows cover 85% of the surface with the remainder terminated by CH3 and H. Subsequent dosing begins a second layer of Al, while the CH3 and H remain trapped at the interface.

FIG. 7. STM image of the Si(001) surface after exposure to DMAH at 700 K. Dim dimers, Al dimer rows, and Si dimer rows are observed. Empty state image, +2 V bias.
Dosing at 700 K, CH3 decomposition is possible and the hydrogen and carbon produce new dim dimer features.13,14 Along with the Al dimers, dense and dilute Si dimers rows are observed. Examples of these are shown in Fig. 7. We presume that these Si atoms have diffused from step edges.15 After continued deposition, after the first layer is saturated, Al forms clusters which cover 40 % of the surface. Thus the decomposition of CH3 leads to increased contamination at the interface.
At room temperature, we observe seven distinct DMAH configurations that display varying degrees of stability against decomposition. We identify these as intact DMAH dimers. A mobile precursor state is observed immediately after the dose. Other metastable states convert or decompose after the surface is annealed to 525 K. However, the dominant configurations (A and D) remain intact. We have identified the predominant dissociation fragments of DMAH: Al, H, and CH3. Pairs of CH3 are observed at the decomposition site while the Al and H diffuse away.
For deposition at 350 K, we observe the initial stages of DMAH decomposition on the surface. The fragments dissociate and form Al dimer rows, CH3, and H after annealing the surface to 525 K.
The optimal temperature range for the cleanest first layer of aluminum is 400 to 575 K. In this temperature range, DMAH is able to dissociate before interacting strongly with the surface (i.e. in configurations A and D). Intact CH3 and H are still observed on the surface.
Dosing above 700 K forms dim dimers, likely resulting from the decomposition of CH3. At higher coverages, Al forms three dimensional clusters, which cover 40 % of the surface. The remaining area is mainly terminated by H.
2 T. Uchiyama and M. Tsukada, J. Vac. Sci. Technol. B 12, 2205 (1994).
3 H. Itoh, J. Itoh, A. Schmid, and T. Ichinokawa, Phys. Rev. B 48, 14663 (1993).
4 G. Brocks, P. J. Kelly, and R. Car, J. Vac. Sci. Technol. B 12, 2705 (1994).
5 Y. Wang, M. J. Bronikowski, and R. J. Hamers, Surf. Sci. 348, 311 (1996).
6 A. J. Mayne, A. R. Avery, J. Knall, T. S. Jones, G. A. g. Briggs, and W. H. Weinberg, Surf. Sci. 284, 247 (1993).
7 A. J. Dyson and P. V. Smith, Surf. Sci. 375, 45 (1997).
8 M. J. Bronikowski and R. J. Hamers, J. Vac. Sci. Technol. A 13, 777 (1995).
9 H. Itoh, J. Itoh, A. Schmid, and T. Ichinokawa, Phys. Rev. B 48, 14663 (1993).
10 C. Zhu, S. Misawa, S. Tsukahara, and S. Fujiwara, Surf. Sci. 357/358, 926 (1996).
11 J. Nogami, A. A. Baski, and C. F. Quate, Phys. Rev. B 44, 1415 (1991).
12 T. Wartik and H. I. Schlesinger, J. Am. Chem. Soc. 75, 835 (1953).
13 M. L. Colaianni, P. J. Chen, H. Gutleben, and J. T. Yates, Chem. Phys. Lett. 191, 179 (1992).
14 L. Li, C. Tindall, O. Takaoka, Y. Hasegawa, and T. Sakurai, Phys. Rev. B/em. 56, 4648 (1997).
15 C. Pearson, B. Borovsky, M. Krueger, R. Curtis, and E. Ganz, Phys. Rev. Lett. 74, 2713 (1995).
16 B. C. Stipe, M.A. Razaei, and W. Ho, J. Chem. Phys. 107, 6443 (1997).
17 The readier may note the strange appearance of the F configuration in this image. At –2 V sample bias, the bright spot of the feature flips about the adjoining dark dimer. One flips during the scan in this image.
18 In order to identify the features produced by hydrogen on the DMAH dosed Si(001) surface, we dosed atomic hydrogen on the clean surface in the fashion of Boland and compared STM images of these two surfaces taken over the full range of sample biases between ± 3 V. [J. J. Boland, Phys. Rev. Lett. 65, 3325 (1990).] The features we identify with H in this paper were identical to those found on the H dosed surface. Similarly, comparison with images from H studies by other groups groups supported our identification. [X.D. Wang, H. Lu, T. Hashizume, H.W. Pickering, and T. Sakurai, Appl. Surf. Sci. 67, 266 (1993) and T. Uchiyama and M. Tsukada, J. Vac. Sci. Technol. B 12, 2205 (1994).]
We would like to thank Craig Moody and Dan Columbo for their assistance and Wayne Gladfelter for valuable discussions. The Schumacher Co. kindly supplied us with the DMAH. We would also like to thank the NSF for supporting this research through grant DMR-9508937.
Jump to
Hot STM Labs
Nanolithography using Chemical
Vapor Deposition and Atomic Hydrogen Resist
Copyright 1998 by the Regents of the University of Minnesota, Dept. of Physics & Astronomy. All rights reserved.